| COOKIES: By using this website you agree that we can place Google Analytics Cookies on your device for performance monitoring. | ![[Talks.cam]](http://talks.cam.ac.uk/images/talkslogosmall.gif?1209136071) |
University of Cambridge > Talks.cam > Theory - Chemistry Research Interest Group > Quantum Chemical Topology: constructing a novel force field with malleable atoms
Quantum Chemical Topology: constructing a novel force field with malleable atomsAdd to your list(s) Download to your calendar using vCal
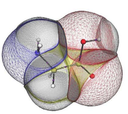
If you have a question about this talk, please contact Robert Best. Quantum Chemical Topology (QCT) [1] is a method [2] that partitions the electron density into so‐called topological atoms, using the insights of dynamical systems. As an example, the figure at top right shows how glycine is partitioned into topological atoms. There are no gaps between these atoms and they do not overlap. Each atom is a box with a particular shape and a finite volume. If the coordinates change then the shapes of the atoms change too, as well as their multipole moments. This complex relationship is captured by machine learning. Topological atoms can be regarded as macroscopic objects, malleable yet open systems. In this talk I will explore how these ideas can be used toenhance the realism of the electrostatic energy a protein force field [3], including polarisation and charge transfer. [1] (a) Popelier, P.L.A.; Bremond,E.A.G. Int.J.Quant.Chem. 2009, 109, 2542. (b) Popelier, P.L. A.; Aicken, F. M. Chem. Phys. Chem. 2003,4, 824. (c) Popelier, P. L. A. Quantum Chemical Topology: on Bonds and Potentials; Springer: Heidelberg, Germany, 2005. [2] (a) Bader, R. F. W. Atoms in Molecules. A Quantum Theor. ; Oxford Univ. Press, 1990. (b) Popelier, P. Atoms in Molecules. An Introduction; Pearson Education: London, 2000. [3] Mills, M. J.L.; Popelier, P. L. A.Theor.Chem.Acc. 2012, 131, 1137. This talk is part of the Theory - Chemistry Research Interest Group series. This talk is included in these lists:
Note that ex-directory lists are not shown. |
Other listsMonitoring Upper Extremity Activity to Track Rehabilitation Progress following Spinal Cord Injury Meeting the Challenge of Healthy Ageing in the 21st Century Nonlinear WavesOther talksAn SU(3) variant of instanton homology for webs Plants of the Richtersveld Regulators of Muscle Stem Cell Fate and Function Adaptive auditory cortical coding of speech Simulating Electricity Prices: negative prices and auto-correlation MOVED TO 28 JUNE 2018 It takes two to tango:platelet collagen receptor GPVI-dimer in thrombosis and clinical implications EU LIFE Lecture - "Histone Chaperones Maintain Cell Fates and Antagonize Reprogramming in C. elegans and Human Cells" Black and British Migration 'Honouring Giulio Regeni: a plea for research in risky environments' The Gopakumar-Vafa conjecture for symplectic manifolds Liver Regeneration in the Damaged Liver Cafe Synthetique: Synthetic Biology Industry Night |